AL Amyloidosis and Other Serious Diseases... Cell Therapy Clinical Trailblazer
Immix Biopharma is a clinical-stage biopharmaceutical company trailblazing cell therapies in AL Amyloidosis and select other serious diseases. Our lead candidate is sterically-optimized BCMA-targeted chimeric antigen receptor T (CAR-T) cell therapy NXC-201. NXC-201 is being evaluated in the U.S.
Latest News
Latest News

Want to Learn More?
Recent Events
Immix Biopharma to Present Abstract at the upcoming ASH 2025 Annual Meeting
12/06/2025 -
12/09/2025
Orlando, Florida
Leerink Partners 2026 Global Healthcare Conference
03/08/2026 -
03/11/2026
Miami Beach, FL
Immix Biopharma to Present Abstract at the upcoming ASH 2025 Annual Meeting
12/06/2025 -
12/09/2025
Orlando, Florida
Immix Biopharma to Present at the 37th Annual Piper Sandler Healthcare Conference
12/03/2025 -
New York, NY
Corporate Highlights
Enrollment Completion of BLA-Enabling Relapsed/Refractory AL Amyloidosis Trial NEXICART-2
Lead site Memorial Sloan KetteringCancer Center (MSKCC) for US AL
Amyloidosis NXC-201 clinical trial.
Immix Biopharma Receives U.S. FDA Breakthrough Therapy Designation for NXC-201
Opens up ability to treat patients in the US. Favorable tolerability enables potential expansion into other serious disease indications.
NXC-201 demonstrated a complete response (CR) rate of 75% (15/20) (at s/u IFE(-) level) by independent review committee at ASH 2025
A Better Solution
NXC-201 phase 2 relapsed/refractory AL Amyloidosis clinical trial NEXICART-2: 75% (up to 95%) complete response rate ($3bn market)
Received FDA Breakthrough Therapy, RMAT and Orphan Drug Designations for NXC-201 for Al Amyloidosis.
We Are on the Brink of
Turning Despair into Hope
Our progress is accelerating toward commercial launch
Founded Immix Biopharma to change the sentence for patients
2022
- Systematic search launched and concluded to identify breakthrough cell therapies
- Licensed-in global rights intellectual property from Hadassah Israel
2023-2024
- FDA Orphan Drug & FDA Designations secured
- NEXICART‑1 ex‑U.S. trial: 75% complete response rate
- Mentioned in NEJM AL Amyloidosis review
Q1 2025
- Published in Journal of Clinical Oncology
Q2 2025
- American Society of Clinical Oncology (ASCO) Oral Presentation
Q4 2025
- American Society of Hematology (ASH) Oral Presentation
- Onboarded Chief Commercial Officer
2026
- Received FDA Breakthrough Therapy Designation
Coming Next
- BLA submission to FDA
- Commercial Launch
Blue Ocean Opportunity
In relapsed/refractory AL Amyloidosis there are no drugs approved today (Amyloidosis is a $3 billion dollar market according to Grand View Research).
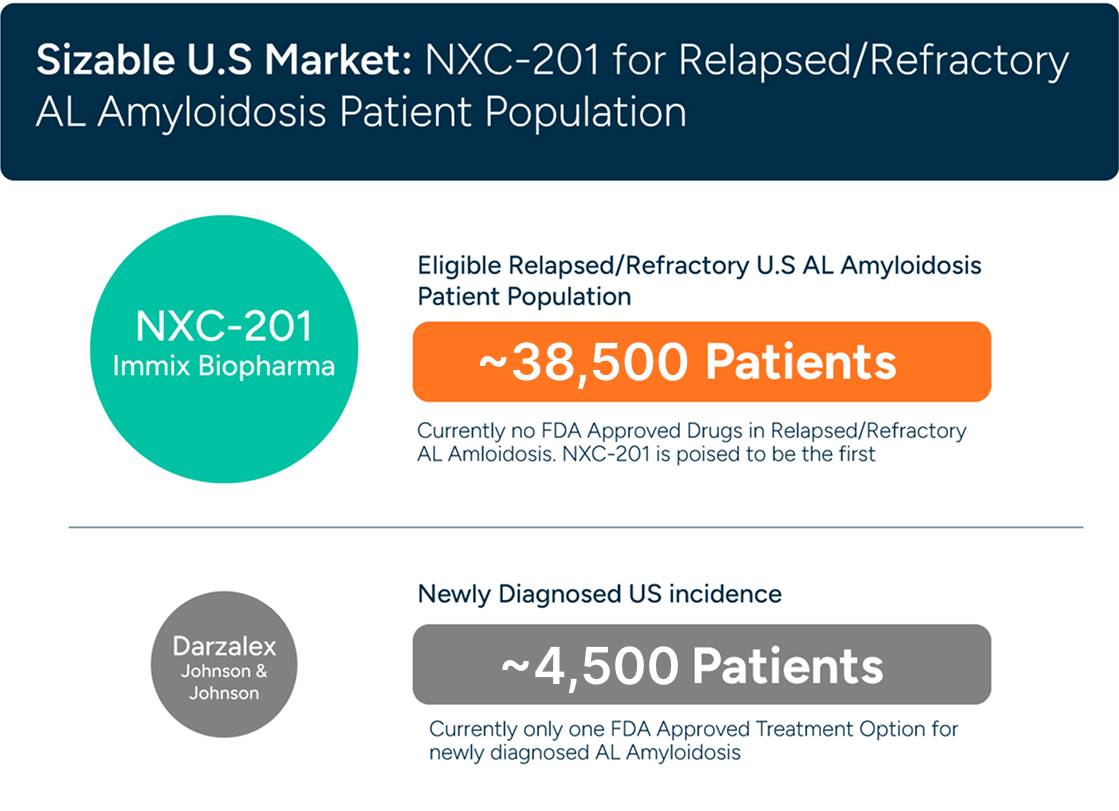
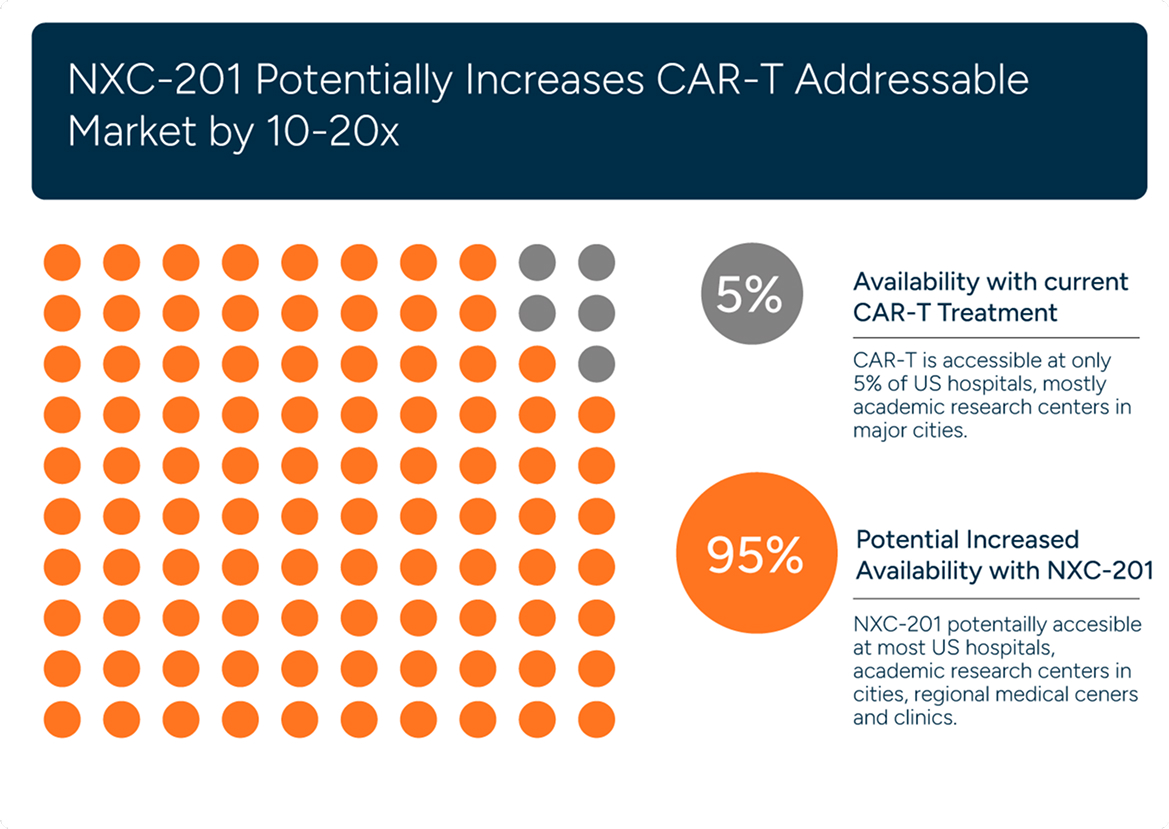
AL Amyloidosis Market
Industry Leading Partners
Immix Biopharma collaborates with top-tier industry leaders to accelerate our research and development efforts. Together, we are committed to advancing groundbreaking therapies that promise to transform patient outcomes in AL Amyloidosis and select other serious diseases.

Beyond AL Amyloidosis: A Platform Built to Transform Multiple Diseases
Immix is laser focused on establishing NXC‑201 as the standard of care for relapsed/refractory AL amyloidosis. Our breakthrough also represents far more than a single‑disease solution.
NXC‑201 targets plasma cells, the antibody factories of the immune system. Success in AL amyloidosis validates our approach and opens the door to treating dozens of immune‑mediated diseases affecting millions of patients worldwide.
Neurology
Rheumatology
Hematology
Vascular & More
$10B+
Total Addressable Market
Across plasma cell-driven immune diseases, with AL amyloidosis alone representing $1.4-2.8 billion in annual sales potential
Analyst Coverage
Morgan Stanley
Judah Frommer
Mizuho Securities
Douglas MacPherson, Ph.D.
Sam Lee, Ph.D.
Ryan Ries, Ph.D
Citizens JMP Securities
Reni Benjamin, Ph.D.
H.C. Wainwright
Robert Burns
SEC Filings
Investor FAQs
Where is Immix Biopharama's corporate headquarters?
Our corporate headquarters are in Los Angeles, California.
Where is Immix Biopharma incorporated?
Immix Biopharma is incorporated under the laws of the State of Delaware.
What is Immix Biopharma's fiscal year?
Our fiscal year is January 1st to December 31st.
How is Immix Biopharma's stock traded?
We have been approved to list our shares of common stock on The Nasdaq Capital Market under the symbol “IMMX” upon our satisfaction of Nasdaq’s initial listing criteria, including the completion of initial public offering.
Does Immix Biopharma pay dividends?
Immix Biopharma has not historically paid any dividends and does not expect to pay dividends in the foreseeable future.
Who is Immix Biopharma's transfer agent?
Immix Biopharma’s transfer agent is Philadelphia Stock Transfer (PST)
Whom do I contact with questions about my stock?
You may contact PST at:
Philadelphia Stock Transfer, Inc.
2320 Haverford Road, Suite 230,
Ardmore, PA 19003
(484) 416-3124
Who is Immix Biopharma's auditor?
Crow LLP
Who is Immix Biopharma's legal counsel?
Sheppard, Mullin, Richter & Hampton LLP
30 Rockefeller Plaza
New York, NY 10112
T: +1.212.653.8700
F: +1.212.653.8701
Where can I find out more about Immix Biopharma?
Additional information about Immix Biopharma can be found on our website and in our public filings with the United States Securities & Exchange Commission.
Where can I get the latest corporate news releases and financial reports?
Our latest corporate news releases and financial reports can be found on the Investors page of our website.
How can I view documents Immix Biopharma has filed with the Securities & Exchange Commission (SEC)?
These documents can be found on the SEC’s EDGAR system at www.sec.gov or on the Investors page of our website.
Who makes up the Executive Management team and Board of Directors?
site.
How can I obtain further information or materials on Immix Biopharma?
You can contact us by using the Contact Us page on our website.
